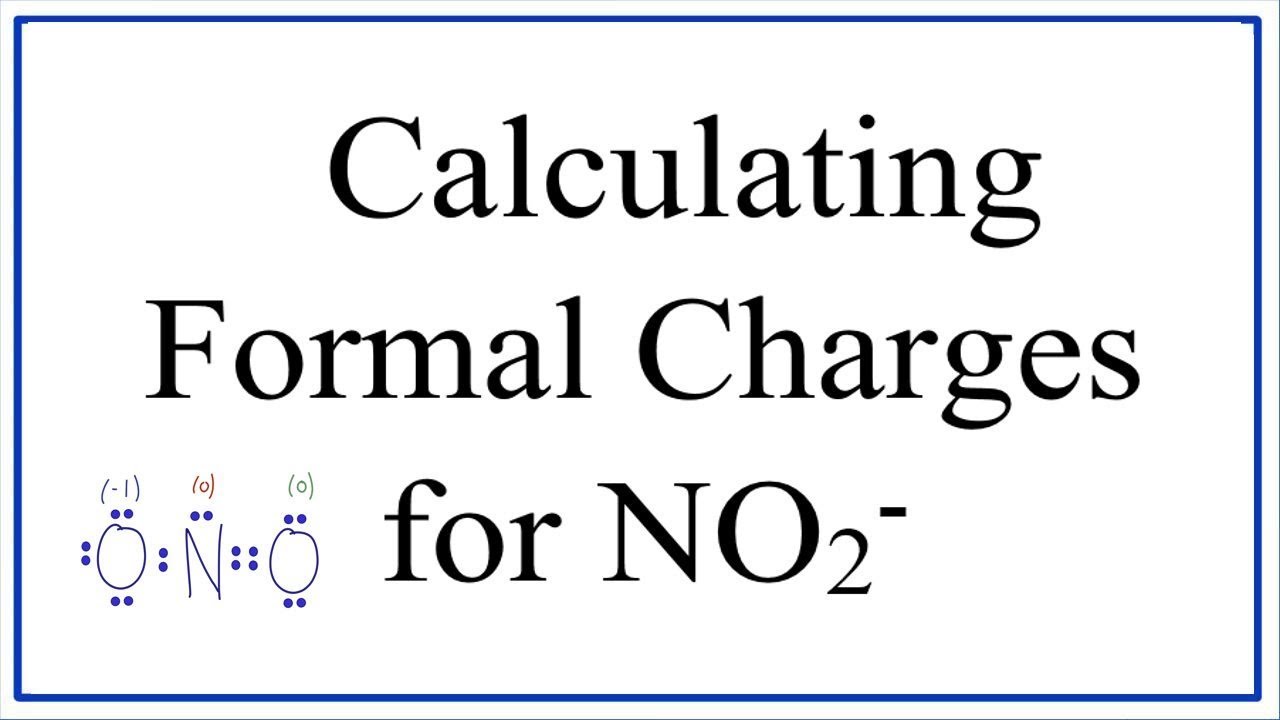
Therefore, the shape of a molecule is determined by the number of lone pairs present as well as the extent of electronic repulsion present in the molecule.Īlso, the lone pair of electrons present upon the central atom determines the distortion of the bond angle between the central atom and other atoms. However, it is the maximum between lone pairs of electrons due to the available free space around them. This repulsion exists between bonding as well as non-bonding electrons (lone pairs). The Valence Shell Electron Pair Repulsion (VSEPR) Theory clearly states that electrons inside a molecule tend to arrange themselves in a manner to avoid inter-electronic repulsion. 
Therefore, the total formal charge on the molecule also becomes zero indicating that the derived Lewis structure is correct. Putting these values in above formula, Formal Charge = 7 – = 0
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
